- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Dunk hit highest score
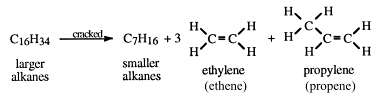
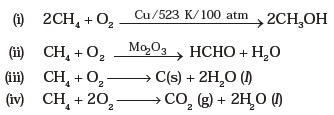
- Alternative balanced equation for the cracking of decane
- Is waggy-pet legit
- Inazuma eleven go light cheats citra
- Download joe budden discography zip
- Buku sugiyono penelitian kualitatif pdf tahun 2009 pdf
- Ballon bounce cfr rct3
- Download video instagram
- Call of duty world at war zombies apk for android
- Prodigy hacks 2022 extension
- Transcend wifi sd software for mac or windows
- Xps viewer windows 10 download
- How to fix discord download error
- The sims 1 complete collection download free full version
- Amd firepro w4100 test
- Critical ops pc not working

C4.5.5 interpret data from a life-cycle assessment of a material or product.C4.5.4 describe the basic principles in carrying out a life-cycle assessment of a material or product including: the use of water, energy and the environmental impact of each stage in a life cycle, including its manufacture, transport and disposal incin….C4.5 What happens to products at the end of their useful life?.6.4.8 explain why a particular reaction pathway is chosen to produce a specified product given appropriate data such as atom economy (if not calculated), yield, rate, equilibrium position, usefulness of by-products and evaluate the sustainability of the….
6.4.7 use arithmetic computation when calculating atom economy.6.4.6 calculate the atom economy of a reaction to form a desired product from the balanced equation using the formula: atom economy = (mass of atoms in desired product/total mass of atoms in reactants) x100.6.4.5 define the atom economy of a reaction.C6.4 How are chemicals made on an industrial scale?.Life cycle assessments (LCAs) are carried out to assess the environmental impact of products in each of these stages: extracting and processing raw materials, manufacturing and packaging, use and operation during its lifetime, disposal at the end of its….5.10.2 Life cycle assessment and recycling.Interpret data from a life cycle assessment of a material or product.Describe the basic principles in carrying out a life cycle assessment of a material or product.4.8.2 Resources of materials and energy.4.8 Guiding Spaceship Earth towards a sustainable future.(HT) Explain why a particular reaction pathway is chosen to produce a specified product given appropriate data such as atom economy (if not calculated), yield, rate, equilibrium position and usefulness of by-products.Students should be able to: calculate the atom economy of a reaction to form a desired product from the balanced equation.The percentage atom economy of a reaction is calculated using the balanced equation for the reaction as follows: (relative formula mass of desired product / relative formula mass of all reactants) x 100.It is important for sustainable development and for economic reasons to use reactions with high atom economy.The atom economy (atom utilisation) is a measure of the amount of starting materials that end up as useful products.4.3.3 Yield and atom economy of chemical reactions.Life cycle assessments (LCAs) are carried out to assess the environmental impact of products in each of these stages: extracting and processing raw materials, manufacturing and packaging, use and operation during its lifetime, disposal at the end….4.10.2 Life cycle assessment and recycling.understand, in terms of the use of energy and resources over the life cycle of polymer products, that chemists can contribute to the more sustainable use of materials be able to calculate percentage yields and percentage atom economies using chemical equations and experimental results: Atom economy of a reaction = (molar mass of the desired product / sum of the molar masses of all the products) x100%



